KEY POINTS
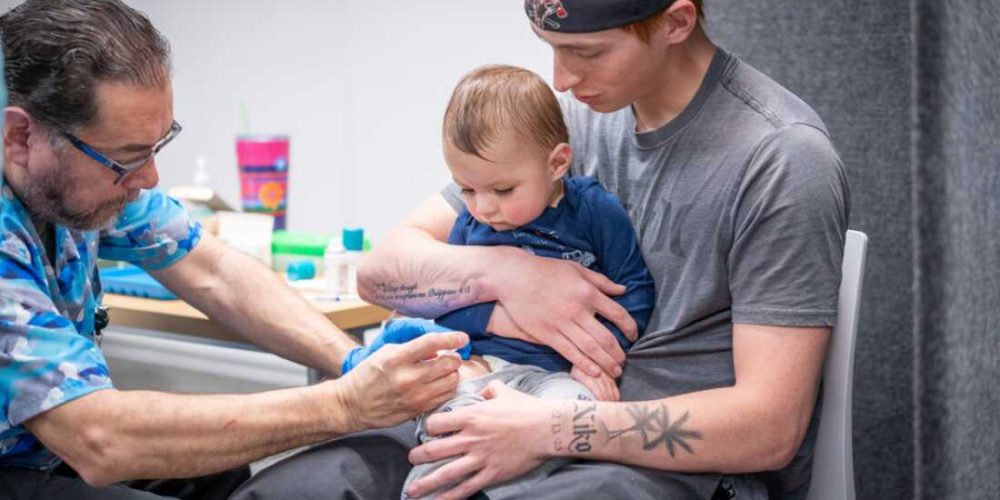
- U.S. District Judge Brian Murphy has issued a preliminary injunction blocking key parts of Health Secretary Robert F. Kennedy Jr.’s plan to drastically reduce recommended childhood vaccinations.
- The ruling halts the implementation of a revised January schedule that cut the number of routinely recommended shots from 17 to 11 and downgraded protections for diseases like flu and hepatitis A.
- The judge also suspended 13 of Kennedy’s appointees to the Advisory Committee on Immunization Practices (ACIP), declaring the panel “unlawfully constituted” and lacking the scientific expertise required by law.
A federal judge in Boston has delivered a significant legal setback to the Trump administration’s efforts to reshape American public health, temporarily blocking a series of controversial changes to the national childhood immunization schedule. In a 45-page ruling, Judge Brian Murphy sided with the American Academy of Pediatrics (AAP) and other major medical groups, concluding that the Department of Health and Human Services (HHS) likely violated federal procedures and disregarded established scientific methods.
The court’s decision effectively restores the vaccine recommendations that were in place prior to January 5, 2026. This means the broad federal guidance once again includes routine vaccinations for influenza, rotavirus, hepatitis A, hepatitis B, RSV, and certain types of meningitis for all children—recommendations that Secretary Kennedy had sought to end or downgrade to “shared clinical decision-making.” Judge Murphy emphasized that for decades, U.S. policy has been rooted in a “method scientific in nature,” which he argued the current administration had “disregarded.”
A major focal point of the ruling was the Advisory Committee on Immunization Practices (ACIP), the body that shapes insurance coverage and doctor recommendations. Last year, Secretary Kennedy dismissed all 17 independent experts on the panel, replacing them with his own appointees. Judge Murphy noted that most of these new members appeared “distinctly unqualified,” with only six having meaningful experience in vaccine research. Under the Federal Advisory Committee Act, such panels must be “fairly balanced,” a standard the judge ruled was not met by the current group of vaccine skeptics.
As a direct result of the injunction, a high-stakes ACIP meeting scheduled to begin this Wednesday in Atlanta has been postponed. The judge also invalidated several prior votes taken by the reconstituted committee, including their decisions to roll back recommendations for newborn hepatitis B shots and universal COVID-19 vaccinations. However, the ruling was not a total sweep for the plaintiffs; the judge declined to block a separate May order that stops the CDC from recommending COVID-19 shots for healthy children and pregnant women at this time.
The Trump administration has signaled its intent to appeal the decision, with HHS spokespeople characterizing the ruling as “judicial overreach.” Government lawyers argued that the Secretary holds broad authority to adjust policy to address a decline in public trust. Conversely, public health experts and the AAP hailed the decision as a “historic victory” for evidence-based medicine, arguing that it prevents “capricious policy” from harming vaccination rates and public health.
For parents and pediatricians, the ruling brings immediate clarity to a period of intense confusion. While dozens of states had already rejected the administration’s new guidelines in favor of traditional medical advice, the federal stay ensures that the “gold standard” schedule remains the official baseline for insurance and clinical practice while the broader legal battle continues.