KEY POINTS
- Researchers at Osaka University identified the integrin αV-YAP-CTGF signaling pathway as the primary driver of liver tissue scarring.
- The study highlights how physical pressure from stagnant blood flow triggers molecular changes that lead to fibrosis and tumor growth.
- This discovery provides a clear therapeutic target for preventing organ failure in patients with chronic heart and liver conditions.
Medical researchers in Japan have reached a significant milestone in understanding how chronic blood flow issues in the liver evolve into life-threatening conditions. For years, clinicians have observed a strong correlation between liver congestion—a state where blood flow slows or stops—and the development of severe scarring and even malignancy. However, the precise biological bridge between these states remained a mystery until now. A new study from Osaka University has finally mapped the molecular route that leads from simple congestion to advanced organ failure.
The condition, often referred to as congestive hepatopathy, frequently affects individuals with underlying heart issues. It is particularly common in patients who have undergone the Fontan procedure, a complex surgery for congenital heart defects. When the heart cannot pump efficiently, blood backs up into the liver, creating internal pressure. Over time, this stagnation triggers a cascade of biological failures, including portal hypertension, cirrhosis, and eventually liver cancer. By identifying the exact switch that turns on this damage, researchers believe they have found a way to potentially stop the progression of the disease.
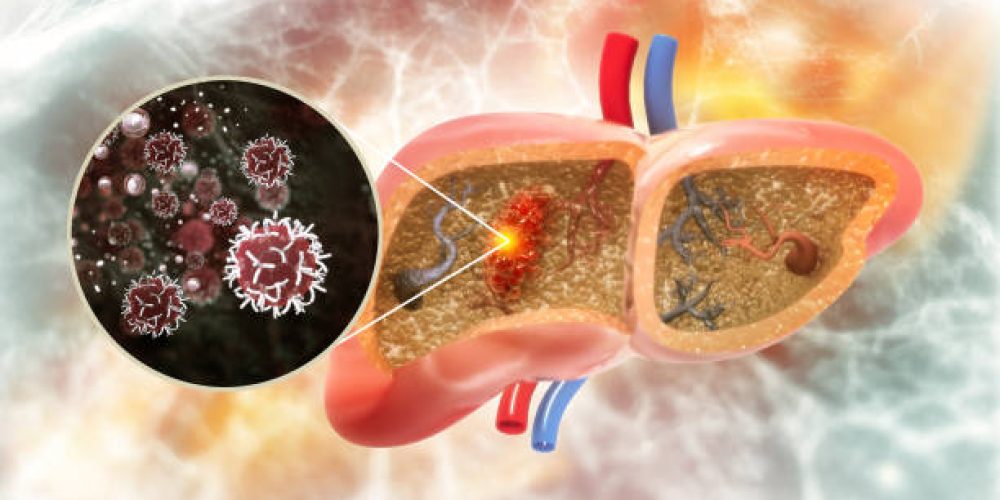
The research team focused their investigation on liver sinusoidal endothelial cells, or LSECs. These specialized cells line the smallest blood vessels within the liver and act as the first point of contact for blood flow. Because they are physically positioned to feel changes in circulation, the researchers suspected they played a key role in sensing the mechanical stress caused by congestion. Using advanced spatial transcriptomics and single-cell analysis, the team compared liver samples from mice and human patients to see how these cells responded to increased pressure.
The data revealed a specific signaling axis involving three primary components: integrin αV, Yes-associated protein (YAP), and connective tissue growth factor (CTGF). When blood flow becomes sluggish, the physical force, known as hydrostatic pressure, activates the integrin αV molecule. This activation then triggers YAP, which in turn causes the cell to overproduce CTGF. It is this specific chemical overproduction that stimulates the growth of fibrous scar tissue and creates an environment where cancerous tumors can take root.
To verify their findings, the scientists tested various intervention methods in laboratory models. They found that by either blocking the integrin αV molecule or removing the CTGF protein from the equation, they could significantly reduce the amount of liver damage. These experiments suggest that the pathway is not just a marker of the disease but the actual engine driving it. By targeting these specific molecules with new drugs, doctors might be able to prevent the liver from scarring even if the underlying blood flow issue cannot be immediately corrected.
The implications of this discovery extend beyond heart patients. The researchers noted that the same high-pressure environment found in liver congestion is also present in patients suffering from general liver cirrhosis. This means that a treatment developed for congestive hepatopathy could potentially help millions of people with various forms of chronic liver disease. The study provides a clear target for future drug development, shifting the focus from managing symptoms to interrupting the cellular signals that cause the organ to fail.
Ultimately, this breakthrough provides hope for a more proactive approach to liver health. Instead of waiting for fibrosis to reach a critical stage, medical professionals may soon have the tools to intervene at the molecular level the moment congestion is detected. By silencing the integrin αV-YAP-CTGF pathway, it may be possible to keep the liver functional and cancer-free, even in the face of chronic circulatory challenges.