KEY POINTS
- Several major Indian drug manufacturers have officially released generic versions of semaglutide to compete with high-cost international brands.
- These domestic alternatives are expected to reduce the monthly cost of treatment by nearly 70%, significantly increasing accessibility for patients.
- Regulatory approval follows the expiration of specific local patent protections, allowing India to leverage its massive manufacturing scale for global distribution.
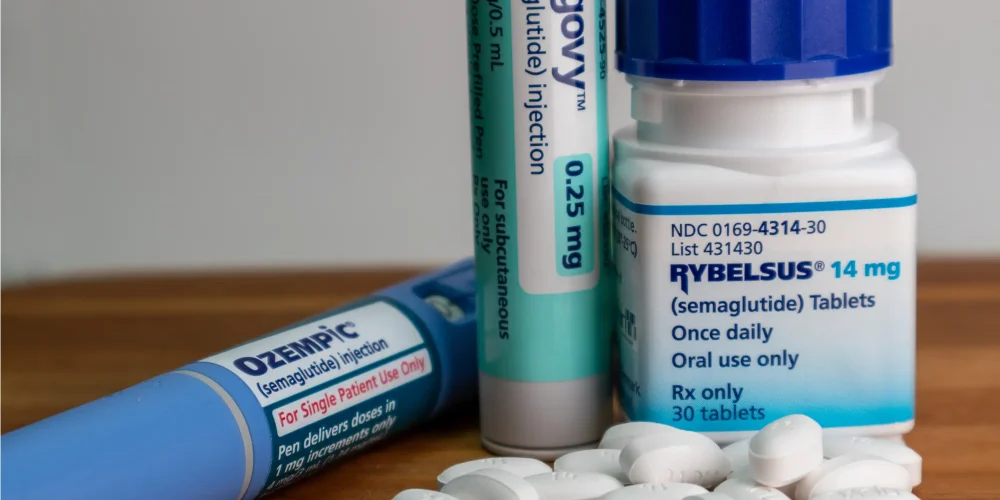
The landscape of metabolic healthcare is undergoing a radical shift as Indian pharmaceutical companies begin flooding the market with cost-effective versions of popular weight-loss drugs. For years, the global supply of semaglutide—the active ingredient in medications like Ozempic and Wegovy—was tightly controlled and priced at a premium. However, a wave of new domestic launches in India is now providing patients with equivalent treatments at a fraction of the previous cost. This surge in production marks a pivotal moment for the industry, as India utilizes its reputation as the pharmacy of the world to address the global obesity and diabetes crisis.
Market analysts observe that the entry of these generic competitors will likely disrupt the pricing strategies of multinational pharmaceutical corporations. While original manufacturers have struggled with supply chain shortages and high retail prices, Indian firms like Sun Pharma and Dr. Reddy’s are scaling up production rapidly. By offering these life-changing medications at significantly lower price points, these companies are targeting a vast demographic of middle-income patients who were previously priced out of the market. This increased competition is expected to force a broader reassessment of how weight-loss drugs are distributed and covered by insurance providers globally.
The regulatory environment in India played a crucial role in this market expansion. Recent legal clarifications regarding patent durations have opened the door for local scientists to refine their own bio-equivalent versions of the drug. These new products have undergone rigorous testing to ensure they meet safety and efficacy standards comparable to the original formulas. Health officials emphasize that while the price is lower, the quality of the medication remains high due to the advanced manufacturing technologies employed by India’s leading labs. This balance of affordability and reliability is a hallmark of the Indian pharmaceutical sector’s global strategy.
Beyond the immediate impact on individual patients, the influx of cheaper semaglutide has significant implications for public health systems. Governments in developing nations are now looking at these generic options as a viable way to manage rising rates of type 2 diabetes and obesity-related complications. By lowering the financial barrier to entry, healthcare providers can implement larger-scale intervention programs. This shift could lead to a long-term reduction in the strain on hospitals and clinics, as more people gain access to preventative treatments that manage their metabolic health effectively before chronic issues arise.
Looking forward, the global pharmaceutical industry is bracing for a “generic wave” that could extend far beyond the borders of South Asia. As these Indian-made versions gain international certifications, they are likely to be exported to markets in Africa, Southeast Asia, and parts of Europe. This expansion will challenge the dominance of Western drugmakers and could lead to a more democratized landscape for essential medicines. For now, the focus remains on meeting the massive domestic demand in India, but the ripple effects of this production surge will undoubtedly be felt across the global healthcare economy for years to come.